Smart Drug Registration Made Simple
OntoPharma analyzes your drug composition and identifies licensing requirements across multiple countries, ensuring compliance with a single click.
Why OntoPharma
OntoPharma streamlines the complex drug registration process, saving pharmaceutical companies time and resources while ensuring regulatory compliance.
Regulatory Compliance
Ensure all your drug submissions meet the latest regulatory requirements across multiple jurisdictions.
Time Savings
Reduce research time by up to 95% with our AI-powered analysis of global regulatory requirements.
Cost Efficiency
Minimize expenses related to regulatory consultants and reduce costly submission errors.
Global Reach
Access regulatory requirements for over 40 countries from a single, intuitive platform.
How It Works
-
1
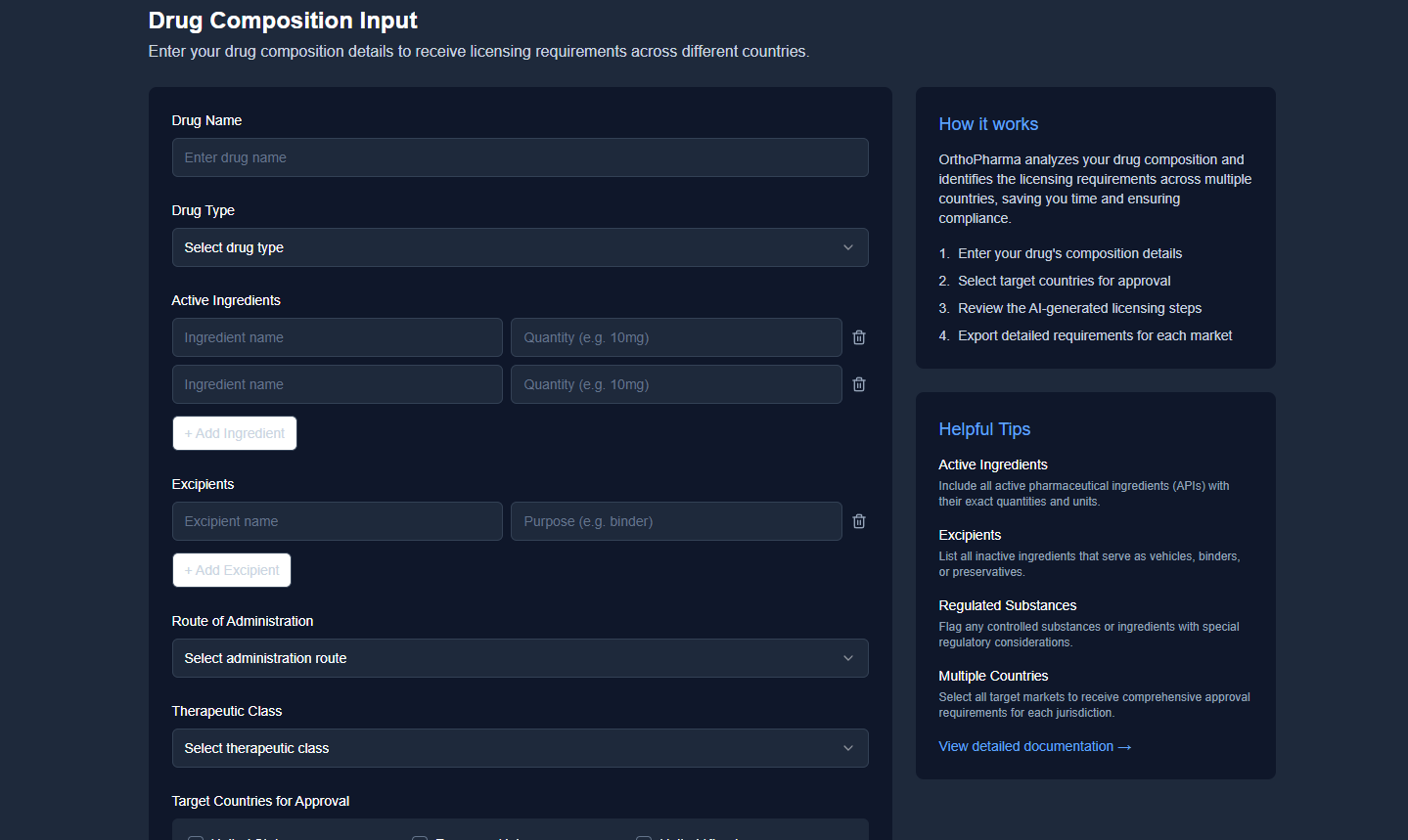
Enter Drug Composition
Input your drug's active ingredients, excipients, and key properties.
-
2
Select Target Countries
Choose which markets you plan to enter with your pharmaceutical product.
-
3
Receive Detailed Requirements
Get customized regulatory steps and documentation needs for each market.
-
4
Export Complete Analysis
Download comprehensive reports for your regulatory teams and stakeholders.

 Chat on LinkedIn
Chat on LinkedIn